
2024 European Innovation Score board Released
2024 European Innovation Score board Released Marie LatourSettings The European Commission released the Innovation Scoreboard 2024 on July 8, offering an in-depth comparative analysis of
Read article at The Smart city journal
Read article at Ticpymes
Read article at Haycanal
Read article at Eleconomista
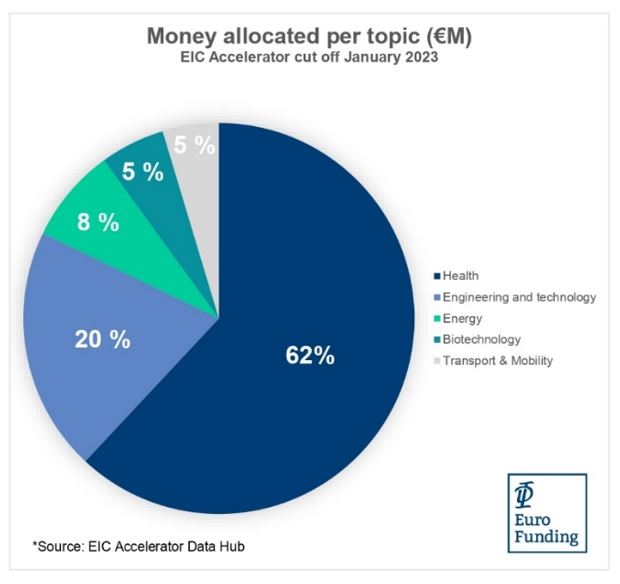
The results of the first European Innovation Council Accelerator cut-off of 2023 are out and they have confirmed an exciting trend with nearly 50% of funded companies and 62% of the overall allocated budget focusing on healthcare innovations. This highlights the interest that this funding program has in driving innovation in the health sector.
The EIC Accelerator provides the financial support that health startups need to bring their products to the forefront of the industry. One of the key benefits of the EIC Accelerator is its ability to provide substantial financial support to startups, allowing them to further develop their technologies and products. The programme offers both grants and equity financing, with the potential for funding up to €17.5 million. This level of financial support is especially useful for startups that may face unique challenges in the market:
Carry out clinical trials: Health startups often need to conduct clinical trials to prove the safety and efficacy of their products. These trials can be very expensive, particularly if they involve many participants or are conducted over a long period of time. Funding from the EIC Accelerator can help cover the costs of these trials, including subcontracting a CRO.
The battle against cancer remains a critical area of research in modern medicine and is a top priority for the European Union, with EU’s Horizon Europe Mission for Cancer aiming to save more than three million lives by 2030 and with a focus on improving the quality and duration of cancer patients’ lives.
Thanks to advancements in technology and biology, doctors and scientists have developed new treatments and therapies that have increased survival rates for cancer patients. However, there is still a long way to go in terms of finding a cure, which is why the EIC Accelerator has launched a new challenge in 2023 to support the development of innovative biomarker-based assays for personalized cancer treatment. The goal of the “Novel biomarker-based assays to guide personalised cancer treatment” challenge is to support health startups working on the development of biomarkers that enable personalized cancer treatment while minimizing adverse effects and improving clinical monitoring.
The EIC has set a budget of €65M for 2023 to fund the development of predictive, prognostic, and companion diagnostic assays to guide cancer treatment, which are in the preclinical or clinical phase 1 stage. By supporting these start-ups in their way to the market, the EIC is helping to advance personalised medicine in the field of oncology and bring society closer to finding a cure for this devastating disease.

2024 European Innovation Score board Released Marie LatourSettings The European Commission released the Innovation Scoreboard 2024 on July 8, offering an in-depth comparative analysis of

Strategic partnership with CleanTech Business Club Marie LatourSettings Joins Forces to Propel Sustainable Innovation On the occasion of its 10th anniversary celebrated in Munich on

2024 European Calls for Environmental and Climate Projects Asier PollánSettings WHAT IS THE PURPOSE OF THIS PROGRAMME? The 2024 LIFE calls for proposals opened on
